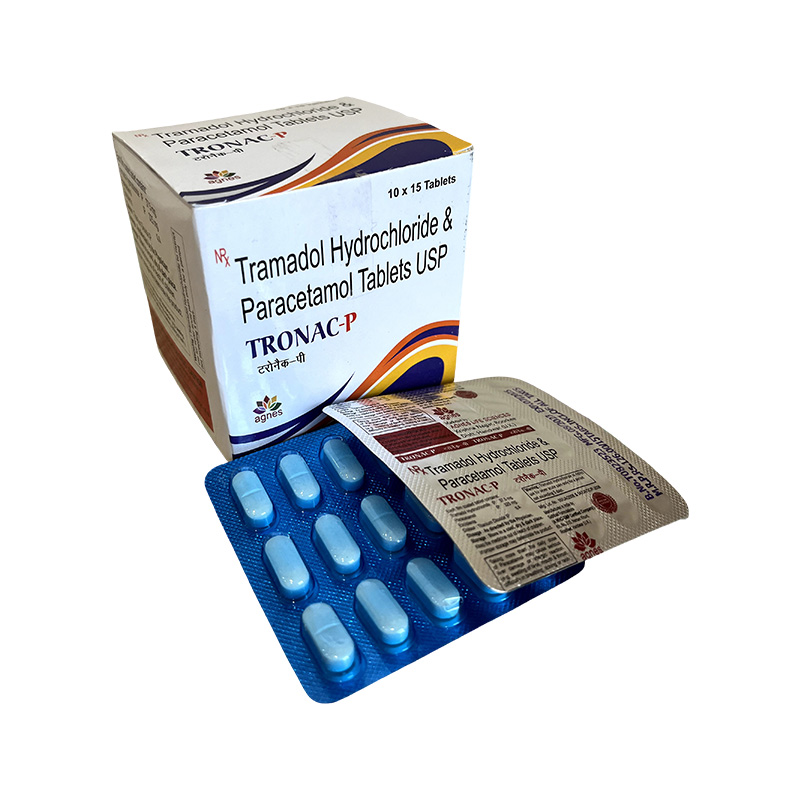
TRONAC-P
Tramadol HCl 37.5mg + Paracetamol 325mg
Category
Tablets
| Packing |
|---|
ENQUIRY
Indications
Moderate to Severe Acute Pain: Specifically indicated for the short-term management of intense pain that does not respond to simple analgesics alone.
Post-Surgical Pain Management: Used to provide effective relief during the recovery phase following major or minor operative procedures.
Traumatic Injury Relief: Targeted for the management of severe pain resulting from fractures, dislocations, or significant soft tissue trauma.
Severe Musculoskeletal Pain: Providing systemic relief for acute flare-ups of lower back pain, sciatica, or joint-related distress.
Dental Pain: Indicated for the management of intense pain following complex dental extractions or oral surgeries.
Post-Surgical Pain Management: Used to provide effective relief during the recovery phase following major or minor operative procedures.
Traumatic Injury Relief: Targeted for the management of severe pain resulting from fractures, dislocations, or significant soft tissue trauma.
Severe Musculoskeletal Pain: Providing systemic relief for acute flare-ups of lower back pain, sciatica, or joint-related distress.
Dental Pain: Indicated for the management of intense pain following complex dental extractions or oral surgeries.
Uses
Dual-Action Pain Suppression: TRONAC-P combines two powerful agents that work on different pathways in the central nervous system to provide superior pain blockade compared to single-ingredient medicines.
Rapid Onset of Relief: The Paracetamol component acts quickly to lower pain signals and reduce fever, while the Tramadol provides a stronger, sustained analgesic effect.
Synergistic Analgesia: By using two different mechanisms, the medication can achieve better pain control with a lower dose of each individual ingredient, improving the overall safety profile.
Enhanced Physical Function: By effectively managing severe pain, patients to begin physical therapy and mobility exercises sooner, accelerating the recovery process.
Interruption of Pain Signals: Specifically targets the “mu-opioid” receptors and inhibits the reuptake of neurotransmitters (serotonin and norepinephrine) to change how the brain perceives pain.
Rapid Onset of Relief: The Paracetamol component acts quickly to lower pain signals and reduce fever, while the Tramadol provides a stronger, sustained analgesic effect.
Synergistic Analgesia: By using two different mechanisms, the medication can achieve better pain control with a lower dose of each individual ingredient, improving the overall safety profile.
Enhanced Physical Function: By effectively managing severe pain, patients to begin physical therapy and mobility exercises sooner, accelerating the recovery process.
Interruption of Pain Signals: Specifically targets the “mu-opioid” receptors and inhibits the reuptake of neurotransmitters (serotonin and norepinephrine) to change how the brain perceives pain.
Dosage & Storage
Standard Dosage: TRONAC-P must be taken exactly as prescribed by your physician. The typical dose is 1 to 2 tablets every 4 to 6 hours as needed for pain, not exceeding the maximum daily limit.
Short-Term Use: This medication is generally intended for short-term use (typically 5 days or less) to manage acute pain episodes.
Storage Conditions: Store TRONAC-P in a cool, dry place, ideally at a temperature below 30°C.
Packaging Integrity: Keep the tablets in their original blister packaging to protect them from moisture and humidity.
Restricted Access: This is a powerful medication. It must be kept in a secure, locked location well out of the reach of children and unauthorized individuals.
Short-Term Use: This medication is generally intended for short-term use (typically 5 days or less) to manage acute pain episodes.
Storage Conditions: Store TRONAC-P in a cool, dry place, ideally at a temperature below 30°C.
Packaging Integrity: Keep the tablets in their original blister packaging to protect them from moisture and humidity.
Restricted Access: This is a powerful medication. It must be kept in a secure, locked location well out of the reach of children and unauthorized individuals.
Directions For Use
Administration: Swallow the tablet whole with a full glass of water. It can be taken with or without food, though taking it with food may help reduce the risk of nausea.
Avoid Alcohol: Strictly avoid the consumption of alcohol while taking TRONAC-P, as it can dangerously increase the sedative effects and risk of liver damage.
Consistency: For the best management of severe pain, take the medicine at the specific intervals recommended by your doctor rather than waiting for the pain to become unbearable.
Safe Disposal: If you have leftover medication after your pain has resolved, consult your pharmacist regarding the safe disposal of unused tablets.
Avoid Alcohol: Strictly avoid the consumption of alcohol while taking TRONAC-P, as it can dangerously increase the sedative effects and risk of liver damage.
Consistency: For the best management of severe pain, take the medicine at the specific intervals recommended by your doctor rather than waiting for the pain to become unbearable.
Safe Disposal: If you have leftover medication after your pain has resolved, consult your pharmacist regarding the safe disposal of unused tablets.
Precautions
Liver Health: Since it contains Paracetamol, do not take any other medications containing Paracetamol simultaneously. Excessive intake can cause severe liver damage.
Respiratory Issues: Inform your doctor if you have asthma or any history of breathing difficulties, as Tramadol can occasionally slow down the breathing rate.
Seizure History: Tramadol should be used with extreme caution in patients with a history of epilepsy or those taking other medications that lower the seizure threshold.
Dependency Risk: As a central-acting analgesic, there is a risk of dependency with prolonged or improper use. Always adhere strictly to the prescribed dose and duration.
Pregnancy & Lactation: This medication is generally not recommended during pregnancy or breastfeeding unless specifically determined as essential by a physician.
Respiratory Issues: Inform your doctor if you have asthma or any history of breathing difficulties, as Tramadol can occasionally slow down the breathing rate.
Seizure History: Tramadol should be used with extreme caution in patients with a history of epilepsy or those taking other medications that lower the seizure threshold.
Dependency Risk: As a central-acting analgesic, there is a risk of dependency with prolonged or improper use. Always adhere strictly to the prescribed dose and duration.
Pregnancy & Lactation: This medication is generally not recommended during pregnancy or breastfeeding unless specifically determined as essential by a physician.
Medical Disclaimer
The information provided regarding TRONAC-P is for educational and informational purposes only and is not intended as medical advice. TRONAC-P contains Tramadol (a Schedule H1 drug) and must only be used under the direct supervision of a licensed healthcare professional. Always consult a doctor for a proper diagnosis and a treatment plan tailored specifically to your health needs. Agnes Life Sciences does not claim that its products can diagnose, treat, or prevent any disease without a medical consultation. Individual results and responses to medication may vary.
Indications
Moderate to Severe Acute Pain: Specifically indicated for the short-term management of intense pain that does not respond to simple analgesics alone.
Post-Surgical Pain Management: Used to provide effective relief during the recovery phase following major or minor operative procedures.
Traumatic Injury Relief: Targeted for the management of severe pain resulting from fractures, dislocations, or significant soft tissue trauma.
Severe Musculoskeletal Pain: Providing systemic relief for acute flare-ups of lower back pain, sciatica, or joint-related distress.
Dental Pain: Indicated for the management of intense pain following complex dental extractions or oral surgeries.
Post-Surgical Pain Management: Used to provide effective relief during the recovery phase following major or minor operative procedures.
Traumatic Injury Relief: Targeted for the management of severe pain resulting from fractures, dislocations, or significant soft tissue trauma.
Severe Musculoskeletal Pain: Providing systemic relief for acute flare-ups of lower back pain, sciatica, or joint-related distress.
Dental Pain: Indicated for the management of intense pain following complex dental extractions or oral surgeries.
Uses
Dual-Action Pain Suppression: TRONAC-P combines two powerful agents that work on different pathways in the central nervous system to provide superior pain blockade compared to single-ingredient medicines.
Rapid Onset of Relief: The Paracetamol component acts quickly to lower pain signals and reduce fever, while the Tramadol provides a stronger, sustained analgesic effect.
Synergistic Analgesia: By using two different mechanisms, the medication can achieve better pain control with a lower dose of each individual ingredient, improving the overall safety profile.
Enhanced Physical Function: By effectively managing severe pain, patients to begin physical therapy and mobility exercises sooner, accelerating the recovery process.
Interruption of Pain Signals: Specifically targets the “mu-opioid” receptors and inhibits the reuptake of neurotransmitters (serotonin and norepinephrine) to change how the brain perceives pain.
Rapid Onset of Relief: The Paracetamol component acts quickly to lower pain signals and reduce fever, while the Tramadol provides a stronger, sustained analgesic effect.
Synergistic Analgesia: By using two different mechanisms, the medication can achieve better pain control with a lower dose of each individual ingredient, improving the overall safety profile.
Enhanced Physical Function: By effectively managing severe pain, patients to begin physical therapy and mobility exercises sooner, accelerating the recovery process.
Interruption of Pain Signals: Specifically targets the “mu-opioid” receptors and inhibits the reuptake of neurotransmitters (serotonin and norepinephrine) to change how the brain perceives pain.
Dosage & Storage
Standard Dosage: TRONAC-P must be taken exactly as prescribed by your physician. The typical dose is 1 to 2 tablets every 4 to 6 hours as needed for pain, not exceeding the maximum daily limit.
Short-Term Use: This medication is generally intended for short-term use (typically 5 days or less) to manage acute pain episodes.
Storage Conditions: Store TRONAC-P in a cool, dry place, ideally at a temperature below 30°C.
Packaging Integrity: Keep the tablets in their original blister packaging to protect them from moisture and humidity.
Restricted Access: This is a powerful medication. It must be kept in a secure, locked location well out of the reach of children and unauthorized individuals.
Short-Term Use: This medication is generally intended for short-term use (typically 5 days or less) to manage acute pain episodes.
Storage Conditions: Store TRONAC-P in a cool, dry place, ideally at a temperature below 30°C.
Packaging Integrity: Keep the tablets in their original blister packaging to protect them from moisture and humidity.
Restricted Access: This is a powerful medication. It must be kept in a secure, locked location well out of the reach of children and unauthorized individuals.
Directions For Use
Administration: Swallow the tablet whole with a full glass of water. It can be taken with or without food, though taking it with food may help reduce the risk of nausea.
Avoid Alcohol: Strictly avoid the consumption of alcohol while taking TRONAC-P, as it can dangerously increase the sedative effects and risk of liver damage.
Consistency: For the best management of severe pain, take the medicine at the specific intervals recommended by your doctor rather than waiting for the pain to become unbearable.
Safe Disposal: If you have leftover medication after your pain has resolved, consult your pharmacist regarding the safe disposal of unused tablets.
Avoid Alcohol: Strictly avoid the consumption of alcohol while taking TRONAC-P, as it can dangerously increase the sedative effects and risk of liver damage.
Consistency: For the best management of severe pain, take the medicine at the specific intervals recommended by your doctor rather than waiting for the pain to become unbearable.
Safe Disposal: If you have leftover medication after your pain has resolved, consult your pharmacist regarding the safe disposal of unused tablets.
Precautions
Liver Health: Since it contains Paracetamol, do not take any other medications containing Paracetamol simultaneously. Excessive intake can cause severe liver damage.
Respiratory Issues: Inform your doctor if you have asthma or any history of breathing difficulties, as Tramadol can occasionally slow down the breathing rate.
Seizure History: Tramadol should be used with extreme caution in patients with a history of epilepsy or those taking other medications that lower the seizure threshold.
Dependency Risk: As a central-acting analgesic, there is a risk of dependency with prolonged or improper use. Always adhere strictly to the prescribed dose and duration.
Pregnancy & Lactation: This medication is generally not recommended during pregnancy or breastfeeding unless specifically determined as essential by a physician.
Respiratory Issues: Inform your doctor if you have asthma or any history of breathing difficulties, as Tramadol can occasionally slow down the breathing rate.
Seizure History: Tramadol should be used with extreme caution in patients with a history of epilepsy or those taking other medications that lower the seizure threshold.
Dependency Risk: As a central-acting analgesic, there is a risk of dependency with prolonged or improper use. Always adhere strictly to the prescribed dose and duration.
Pregnancy & Lactation: This medication is generally not recommended during pregnancy or breastfeeding unless specifically determined as essential by a physician.
Side Effects
Medical Disclaimer
The information provided regarding TRONAC-P is for educational and informational purposes only and is not intended as medical advice. TRONAC-P contains Tramadol (a Schedule H1 drug) and must only be used under the direct supervision of a licensed healthcare professional. Always consult a doctor for a proper diagnosis and a treatment plan tailored specifically to your health needs. Agnes Life Sciences does not claim that its products can diagnose, treat, or prevent any disease without a medical consultation. Individual results and responses to medication may vary.